
The mechanisms through which birds are protected against Salmonella Enteritidis are one of the key considerations for developing effective vaccines. Live attenuated and inactivated vaccines against avian Salmonellosis achieve different rates of protection, related mainly to the different immune responses induced.
Live vaccines can stimulate both cellular and humoral responses, while inactivated vaccines, even when generating high antibody titers, fail to induce a cellular response, which is crucial in controlling the pathogen
It is important to study the different roles of the immune response against avian Salmonellosis and to answer the following questions:
- Is humoral immunity sufficient?
- What role do cells play as a defense against salmonellosis?
- How can a vaccine act by exerting competitive exclusion?
Different studies have shown that humoral (antibody production) and cellular (activation of macrophages and lymphocytes) immune responses have a different role in the protection of birds against Salmonella. The humoral response appears to be less essential and is limited to the intestinal level, where IgA (immunoglobulin A) may play a certain role in mucosal protection. On the other hand, recent studies consider that the cellular response is fundamental for the elimination of Salmonella, specifically the response mediated by Th1 instead of Th2.
Is humoral immunity enough?
Antibody levels in the intestine, which reflect the immune response at the mucosal level more accurately than serum antibodies, have been analyzed from biliary samples and there is evidence that animals vaccinated with a live vaccine against Salmonella Enteritidis (PRIMUN SALMONELLA E) and subsequently infected have a high response at the mucosal level, producing IgA antibodies. In contrast, non-vaccinated control animals barely show a response at biliary IgA level. The graph shows that, after being infected, the humoral response is triggered at the local level in vaccinated animals. The presence of IgA at the level of the intestinal mucosa would reduce bacterial adherence and invasion of the intestine.
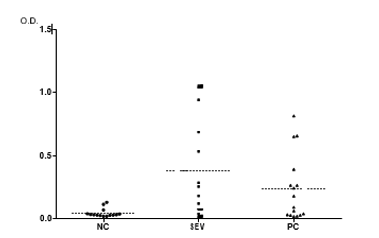
Fig 1. Optical densities (OD) obtained by ELISA test of IgA of biliary samples collected from birds vaccinated on day 1 and challenged on day 14 (the samples are 28 days old). (SEV: group vaccinated with PRIMUN SALMONELLA E; PC: group vaccinated with reference vaccine; NC: non-vaccinated animals). The ELISA test showed a greater biliary IgA response against Salmonella Enteritidis antigens in vaccinated animals (p = 0.01), especially with the PRIMUN SALMONELLA E vaccine
What happens at the cellular level in the intestine?
The intestinal barrier is chickens' first line of defense against Salmonella invasion. The recruitment of immune cells in this organ may be an initial indicator of protection in immunized or non-immunized birds. Most studies focus on cell populations in the blood, but few studies have determined the distribution of immune cells in internal organs with associated lymphoid tissue. The analysis of an intestinal fragment where Peyer's patches are present can reflect what is happening in the target points that are key for Salmonella infection, as can an examination of the population in the cecal tonsils, or analysis of the expression of cytokines at local level.
The cellular immune response in the intestine is highly complex. On the one hand, macrophages have different functions in the early innate immune response against Salmonella. These cells engulf bacteria, present antigens to B and T cells, and produce proinflammatory cytokines that activate other cells of the immune response. The population of macrophages in the intestine has been found to be increased in birds after Salmonella infection. Subsequently, the population of T cells also increases. In general, CD8 + T cells are expected to lead a protective Th1 cell response against secondary Salmonella infection, as has been seen in different studies where this population increased both in the intestine and in the cecal tonsils after primary infection in birds. Similarly, the increase in CD8+ T lymphocytes in cecal tonsils has been correlated with decreased Salmonella counts in the cecum.
These population changes are reflected in the production of inflammatory cytokines and in the recruitment of immune cells in the intestine, meaning cellular immunity can be studied by analyzing the changes in cell populations after vaccination and/or infection (by flow cytometry), or by analyzing the production of cytokines.
The relative expression of cytokines at the level of the ileal mucosa was studied 4 days after vaccination with PRIMUN SALMONELLA E, 4 days after the challenge, and 14 days after the challenge, as shown in Figure 2.
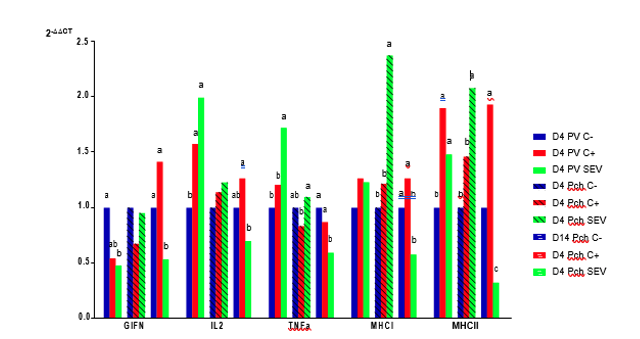
Figure 2: Relative expression at the level of the ileal mucosa of the interferon gamma gene (GIFN), the interleukin 2 gene (IL2), the tumor necrosis factor alpha gene (TNFα), the major histocompatibility complex class I (MHCI) gene, and the major histocompatibility complex class II (MHCII) gene 4 days post-vaccination (D4 PV), 4 days post-challenge (D4 Pch), and 14 days post-challenge (D14 Pch). (SEV: group vaccinated with PRIMUN SALMONELLA E; C +: group vaccinated with the Salmonella Enteritidis reference vaccine; C-: non-vaccinated animals).
Cytokine expression analysis four days after vaccination revealed significant differences between non-vaccinated animals and animals vaccinated with PRIMUN SALMONELLA E. In the case of gamma interferon (GIFN), a significant reduction was observed in the PRIMUN SALMONELLA E vaccine compared to non-vaccinated animals. Additionally, for interleukin 2 (IL2), tumor necrosis factor alpha (TNFα), and major histocompatibility complex class II (MHCII) —not for major histocompatibility complex class I (MHCI)— a positive regulation was observed when compared to non-vaccinated animals. From these results, it can be deduced that the PRIMUN SALMONELLA E vaccine stimulates the different immunological parameters related to the processing and presentation of antigens to induce a correct immune response. This effect is doubly important at certain times, such as the laying period. In the case of the control vaccine, the results were similar to those of the PRIMUN SALMONELLA E vaccine, although in general they were lower.
Four days after the challenge, the gene expression of MHCI and MHCII was significantly higher in animals immunized with the PRIMUN SALMONELLA E vaccine, which represents an increase in cellular activity at the intestinal level in the vaccinated animals. This increase in cellular activity, especially the MHCII gene (which is related to antigen presenting cells) could explain the reduction of Salmonella at the intestinal level and the downregulation of different cytokines when examined 14 days after infection.
The importance of competitive exclusion (CE) in live vaccines
Finally, a further protective effect of live vaccines against Salmonella Enteritidis is competitive exclusion. The attenuated strain applied from the day of hatching prevents other pathogenic strains from colonizing the intestinal tract owing to the previous presence of the vaccine bacteria that has already been established and can be maintained in that environment for a certain period of time. This is another way in which live vaccines control Salmonella colonization. EC may also play an important role in stimulating mucosal immunity, which could contribute significantly to host resistance.
The configuration of a vaccination program must take into account these three types of immunity, which are crucial when it comes to generating proper, long-lasting protection against avian salmonellosis.



